The MODeRN PERIODIC TABLE
BY: DEREK XIE

What is the Periodic Table?
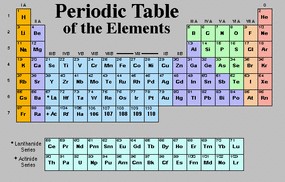
The Periodic Table is a grid that arranges chemical elements according to atomic number. Elements are usually arranged in rows so that elements with similar chemical properties are in vertical columns. The Periodic Table is often used in studying Chemistry.
The Periodic Table is a grid that arranges chemical elements according to atomic number. Elements are usually arranged in rows so that elements with similar chemical properties are in vertical columns. The Periodic Table is often used in studying Chemistry.
|
|
The Story Behind the Periodic Table
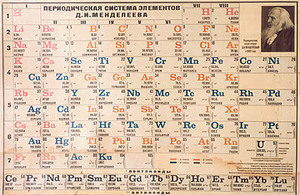
In 1869, Russian scientist (chemist and inventor), Dmitri Mendeleev put together a table that organized elements. Mendeleev was able to put to together a table despite the fact that the Rutherford Model had not yet been invented. The idea of elements did exist at the time and elements had been previously organized however Mendeleev was the first to construct a table similar to the one we have today. The discovery of electrons, protons, and neutrons are only until later however Mendeleev is still able to group elements based on their atomic mass. Perhaps, Mendeleev studied chemical reactions involving large number of atoms to determine atomic mass of certain elements. Mendeleev's hand-written table resembles the modern periodic table in many ways. A few elements were however missing from Mendeleev's table since they had not yet been discovered. On the contrary, Mendeleev's table led to the discovery of many new elements that had not been previously predicted. Such elements include germanium in 1875, gallium in 1879, and scandium in 1886. During Mendeleev's time only over 60 elements are known whereas today over 110 elements are known. The Organization of Elements
In Mendeleev's table, elements were organized according to its atomic weight instead of atomic number. Elements are grouped into rows and columns based on the physical properties. Elements in any column or group (a period) are similar. Elements with a lightest atomic weight in placed in the beginning followed by a heavier element. Hydrogen is the lightest element in comparison to the others. Elements are currently arranged based chemical properties which the number of proton instead of the mass determines the order of the table. The mass is still important since we depend on the atomic masses of element in order to calculate to molar mass of a compound. The organization of the periodic table also contributed to various concepts such as the fact that the period number represent the highest energy level an electron can occupy in the element in the unexcited state( atoms with electrons in the lowest possible energy level) . Contributions in Later Years
Mendeleev was right when he decided to leave spaces blank in his table for undiscovered elements. Many scientist doubted Mendeleev's table at first however the discovery of elements like germanium proved that Mendeleev was correct. Scientists later began to notice that organization based on atomic mass may be a problem since argon is not as reactive as sodium or lithium which are close by in the table. Therefore, the modern periodic table is organized based on atomic number. In 1884, Sir William Ramsay and Lord Rayleigh discovered noble gases which were eventually added to the periodic table as a period 0. The further introduction of man-made elements further expanded the periodic table overtime. Scientists at the time refuse to believe that these radioactive elements are new elements. These elements all go through nuclear fission. In 1913, British physicist Henry Moseley varied that an element's chemical properties are only slightly related to that of its atomic weight and that in reality, it is equal to the number of protons plus the number of neutrons in the nucleus. In 1945, Glenn Seaborg identified lanthanides and actinides and placed them at the bottom of the table. |
Conclusion
Mendeleev's table is considered to be most similar to the table that we use today. Unlike today's table Mendeleev's table lacks certain elements and was arranged according to atomic weight. With improvements from scientists in the 19th and 20th century, new elements were introduced and new rows were added. In the course of overall 100 years the modern periodic table was created.
Mendeleev's table is considered to be most similar to the table that we use today. Unlike today's table Mendeleev's table lacks certain elements and was arranged according to atomic weight. With improvements from scientists in the 19th and 20th century, new elements were introduced and new rows were added. In the course of overall 100 years the modern periodic table was created.