Answers explained
1. Nomenclature
This process sets rules in naming ionic compounds, binary covalent compounds, and acids.
2. Dalton
It is true that Democritus first suggested the existence of the atom but John Dalton supported the idea of the atom as a fundamental chemical object. The theory states that all matter is made of atoms, atoms are indivisible and indestructible, all atoms of a given element are identical in mass and properties, compounds are formed by a combination of two or more different kinds of atoms, and a chemical reaction is a rearrangement of atoms.
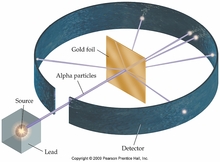
3. Nucleus
Rutherford concluded that all of the positive charge and essentially all of the mass of the atom is concentrated in a small fraction of the total volume of the atom.
4. Empirical
The empirical formula is a formula giving the proportions of the elements present in a compound but not the actual numbers or arrangement of atoms which would otherwise be a molecular formula.
5. Fluorine
Fluorine has 5 electrons in it's 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons, so since Fluorine is so close to ideal electron configuration, the electrons are held very tightly to the nucleus. In other words, Fluorine is so close to completing its ideal electron configuration causing it to have a high attraction for bonding pairs of electrons so that it can complete its configuration.
6. Radius
The nucleus isn't able to attract electrons as well since they are further away and more shielded by other shells that are closer to it which causes the atom to be bigger in size.
7. Resonance
Resonance structures are two or more forms of a molecule where the chemical connectivity is the same but the electrons are distributed differently around the structure (the different position of a double bond). Formal charges are used to find the overall charge and if two forms of a molecule have the same charge with different bond positions, they are resonance structures.
8. Deposition
Deposition is an exothermic process that can be displayed when snow forms in clouds or when frost forms.
9. Limiting
The limiting reactant is the reactant that is completely used up. If there is no longer a reactant, the reaction cannot continue thus ending the reaction as a whole. No more products can be produced if a reactant is missing.
10. Neutralization
In a reaction in water, neutralization results in there being no excess of hydrogen or hydroxide ions present in solution.
11. Collision
The atoms need to collide or come into contact in order to produce a chemical reaction.
12. Calorimetry
Calorimetry measures the heat exchanged with the surroundings.
13. Negative
According to Gibbs Free Energy: ΔG = ΔH - TΔS
One way to look at this question is to plug in values that are known to be favorable. A ΔS that is positive would be favorable because it indicates increasing disorder. A ΔH that is negative would be favorable because it indicates that the reaction is exothermic. So increasing disorder and exothermic will always be spontaneous and with these values, ΔG would be negative.
14. Titration
It is the slow addition of one solution of a known concentration (called a titrant) to a known volume of another solution of unknown concentration until the reaction reaches neutralization, which is often indicated by a color change.
15. Buffer
A buffer solution is typically made by mixing a weak acid and one of its salts OR mixing a weak base with one of its salts. In other words, a buffer solution consists of a mixture of a weak acid and its conjugate base OR a weak base and its conjugate acid.
This process sets rules in naming ionic compounds, binary covalent compounds, and acids.
2. Dalton
It is true that Democritus first suggested the existence of the atom but John Dalton supported the idea of the atom as a fundamental chemical object. The theory states that all matter is made of atoms, atoms are indivisible and indestructible, all atoms of a given element are identical in mass and properties, compounds are formed by a combination of two or more different kinds of atoms, and a chemical reaction is a rearrangement of atoms.
3. Nucleus
Rutherford concluded that all of the positive charge and essentially all of the mass of the atom is concentrated in a small fraction of the total volume of the atom.
4. Empirical
The empirical formula is a formula giving the proportions of the elements present in a compound but not the actual numbers or arrangement of atoms which would otherwise be a molecular formula.
5. Fluorine
Fluorine has 5 electrons in it's 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons, so since Fluorine is so close to ideal electron configuration, the electrons are held very tightly to the nucleus. In other words, Fluorine is so close to completing its ideal electron configuration causing it to have a high attraction for bonding pairs of electrons so that it can complete its configuration.
6. Radius
The nucleus isn't able to attract electrons as well since they are further away and more shielded by other shells that are closer to it which causes the atom to be bigger in size.
7. Resonance
Resonance structures are two or more forms of a molecule where the chemical connectivity is the same but the electrons are distributed differently around the structure (the different position of a double bond). Formal charges are used to find the overall charge and if two forms of a molecule have the same charge with different bond positions, they are resonance structures.
8. Deposition
Deposition is an exothermic process that can be displayed when snow forms in clouds or when frost forms.
9. Limiting
The limiting reactant is the reactant that is completely used up. If there is no longer a reactant, the reaction cannot continue thus ending the reaction as a whole. No more products can be produced if a reactant is missing.
10. Neutralization
In a reaction in water, neutralization results in there being no excess of hydrogen or hydroxide ions present in solution.
11. Collision
The atoms need to collide or come into contact in order to produce a chemical reaction.
12. Calorimetry
Calorimetry measures the heat exchanged with the surroundings.
13. Negative
According to Gibbs Free Energy: ΔG = ΔH - TΔS
One way to look at this question is to plug in values that are known to be favorable. A ΔS that is positive would be favorable because it indicates increasing disorder. A ΔH that is negative would be favorable because it indicates that the reaction is exothermic. So increasing disorder and exothermic will always be spontaneous and with these values, ΔG would be negative.
14. Titration
It is the slow addition of one solution of a known concentration (called a titrant) to a known volume of another solution of unknown concentration until the reaction reaches neutralization, which is often indicated by a color change.
15. Buffer
A buffer solution is typically made by mixing a weak acid and one of its salts OR mixing a weak base with one of its salts. In other words, a buffer solution consists of a mixture of a weak acid and its conjugate base OR a weak base and its conjugate acid.