What is up with Graphite and Diamond?
By: Ellie Ly
So what is the deal with diamond and graphite? Two vastly different materials in color, hardness, uses, and much more. So how is it that two supposedly different materials are both made up of the same element; pure carbon.
First let us take a look at some background on carbon. Carbon is a group 14 non-metal element with an atomic number of 6. At room temperature carbon is a dark grey colored solid, and with a little tweaking, carbon can become diamond and/or graphite.
But how does carbon turn into diamond or graphite?
Well, it is all in the physical structure of the carbon molecules. Elements can have several different structural forms while in the same physical state. These differing forms are known as allotropes.
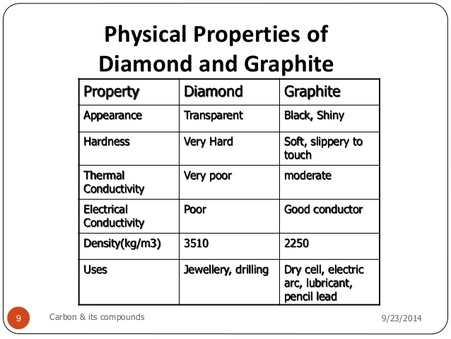
Let us observe the different traits of both diamond and graphite
From the chart it is quite apparent that graphite is a much weaker material than diamond. This is directly correlated to the structure of the carbon molecules in the two materials.
Say you have two buildings.
First let us take a look at some background on carbon. Carbon is a group 14 non-metal element with an atomic number of 6. At room temperature carbon is a dark grey colored solid, and with a little tweaking, carbon can become diamond and/or graphite.
But how does carbon turn into diamond or graphite?
Well, it is all in the physical structure of the carbon molecules. Elements can have several different structural forms while in the same physical state. These differing forms are known as allotropes.
Let us observe the different traits of both diamond and graphite
From the chart it is quite apparent that graphite is a much weaker material than diamond. This is directly correlated to the structure of the carbon molecules in the two materials.
Say you have two buildings.
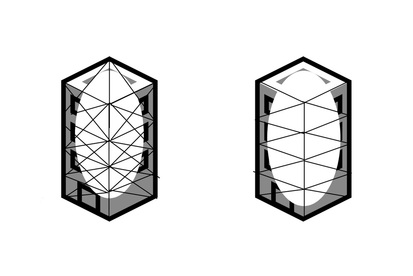
One is built with pillars that are connected and intertwined in a strong fashion while the other building has pillars that are not as connected.
Of the two buildings it is obvious that the one with a more solid structure will be stronger and maybe even prettier than the one with a weaker structure.

This is basically what is happening with the carbon molecules in diamond and graphite!
In diamond, each carbon atom is bonded with four other carbon atoms. These four atoms are again strongly bonded with other four carbon atoms.
Meanwhile in graphite, the carbon atoms form a hexagon shape and each hexagon shape connects to another hexagon. This creates a plane of hexagons; graphite is built by stacking those hexagonal planes on top of one another.
In diamond, each carbon atom is bonded with four other carbon atoms. These four atoms are again strongly bonded with other four carbon atoms.
Meanwhile in graphite, the carbon atoms form a hexagon shape and each hexagon shape connects to another hexagon. This creates a plane of hexagons; graphite is built by stacking those hexagonal planes on top of one another.
As you can see, the carbons in diamond are all connected to each other, while in graphite there are layers of carbon that are not connected. Since the carbons in graphite are not as strongly bounded together as diamond, graphite is a lot weaker than diamond physically.
The question now is how did the carbon molecules form into these two different structures? This is where pressure and temperature comes into play.
As we all know, graphite and diamond are found in the ground, however they are found at different levels of deepness.
Diamond can be found at a deeper level of the earth than graphite. This is because of the difference in pressure and temperature. As you go deeper into the earth, temperature and pressure increases. It takes a higher amount of heat and pressure to create diamond than it does graphite. This is why in games such as Minecraft you can find coal (a low grade graphite) closer to the surface and you can only find diamond at great depths and near lava.
So now you know why diamond and graphite can be totally different but still be made up of the same element.
The question now is how did the carbon molecules form into these two different structures? This is where pressure and temperature comes into play.
As we all know, graphite and diamond are found in the ground, however they are found at different levels of deepness.
Diamond can be found at a deeper level of the earth than graphite. This is because of the difference in pressure and temperature. As you go deeper into the earth, temperature and pressure increases. It takes a higher amount of heat and pressure to create diamond than it does graphite. This is why in games such as Minecraft you can find coal (a low grade graphite) closer to the surface and you can only find diamond at great depths and near lava.
So now you know why diamond and graphite can be totally different but still be made up of the same element.